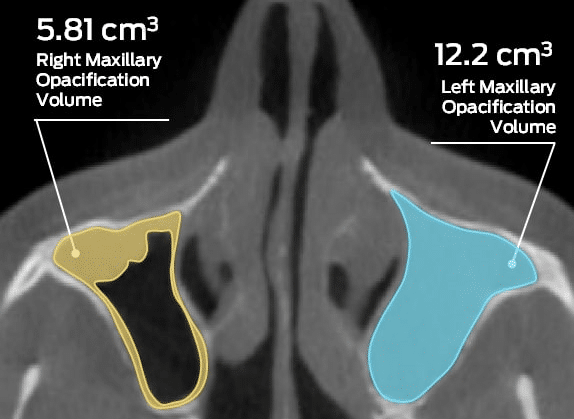
MMI announces global imaging services collaboration with Micron
Learn MoreInsights
Our team is dedicated to staying up to date with the news, discovery, and trends in imaging and clinical trials. Explore what’s new at Medical Metrics and beyond.
MMI News & Events
Something’s always cooking at MMI! From tradeshow attendance to employee promotions, from little joys to major milestones, take a look into what’s happening at our company.
SpineCAMP News
Something’s always cooking at MMI! From tradeshow attendance to employee promotions, from little joys to major milestones, take a look into what’s happening at our company.
LinkedIn Feed
Scientific Resources & Publications
The cornerstone of MMI’s expertise and brilliance lies in the science and technology that make our work possible. Browse our curated resources that form the bedrock of our technical knowledge, as well as internal papers and external publications from partners and clients that we are proud to highlight.
Webinars & Videos
Review our archive of educational webinars, where we engage with luminary key opinion leaders to deliver deep dives into important topics, or peruse our MMI video blog highlighting our services and capabilities.
Industry News
The world of clinical trials is ever-changing, and MMI is devoted to staying up-to-date and integrated with the newest breakthroughs and paradigm shifts. Here we share a selection of articles and thoughts on the ongoing evolution of the clinical trial and imaging fields.
Let MMI provide insights into your clinical study imaging.
Have questions? We’ll connect you immediately to one of our scientific managers and imaging experts. Your time is precious, and we want to make the most out of it.