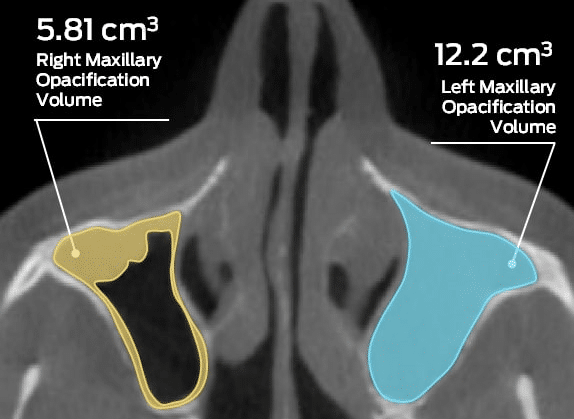
MMI Co-Founder, John Hipp, PhD. to be Guest Editor for Special Issue of Bioengineering! Submit by May 30!
Learn More
Check out MMI's brand new SpineCAMP™ website launch!
Explore SpineCAMP™!
Press Release!: Driving Excellence in Cardiovascular Trials: MMI and Healthcare Inroads Deepen Collaboration
Read HereIndustry News